|
12/30/2023 0 Comments Electron domain geometry of brf3Since there are three fluorine atoms in BrF3, the total number of valence electrons is 7 + 3(7) = 28. To determine the total number of valence electrons in BrF3, we need to consider that bromine contributes 7 valence electrons (since it is in Group 7 of the periodic table) and each fluorine contributes 7 valence electrons. Fluorine, on the other hand, has an atomic number of 9 and an electron configuration of 1s^2 2s^2 2p^5. Bromine has an atomic number of 35, meaning it has 35 electrons. To understand the valence electrons of BrF3, we need to look at the electron configuration of bromine (Br) and fluorine (F). In this article, we will explore the valence electrons and Lewis structure of BrF3, discuss whether it is ionic or covalent, and determine if it is polar or nonpolar. 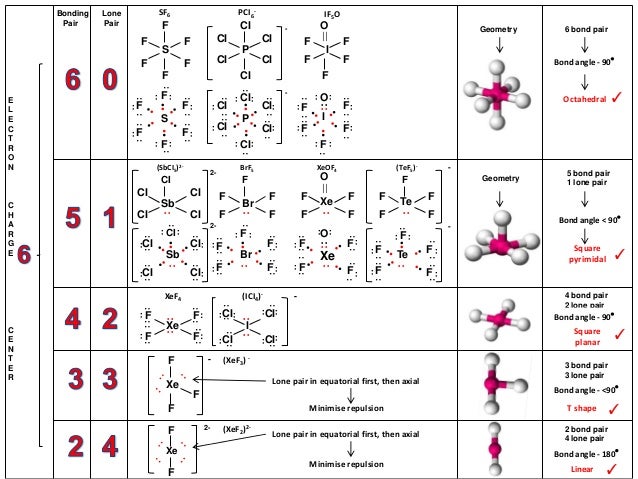 
These concepts help us determine the structure and properties of molecules. In chemistry, understanding the basics of valence electrons, molecular geometry, and chemical bonding is essential. Key Takeaways Atom Number of Valence Electrons Bromine (Br) 7 Fluorine (F) 7 Understanding the Basics The Lewis structure of BrF3 is important in understanding its chemical properties and reactions. This results in a trigonal bipyramidal shape for the molecule. The bromine atom has three lone pairs of electrons, while each fluorine atom has one lone pair. In the BrF3 Lewis structure, there are three fluorine atoms bonded to a central bromine atom. It helps us understand the bonding and shape of the molecule. 
The Lewis structure of BrF3, also known as Bromine Trifluoride, is a diagram that represents the arrangement of atoms and valence electrons in the molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
